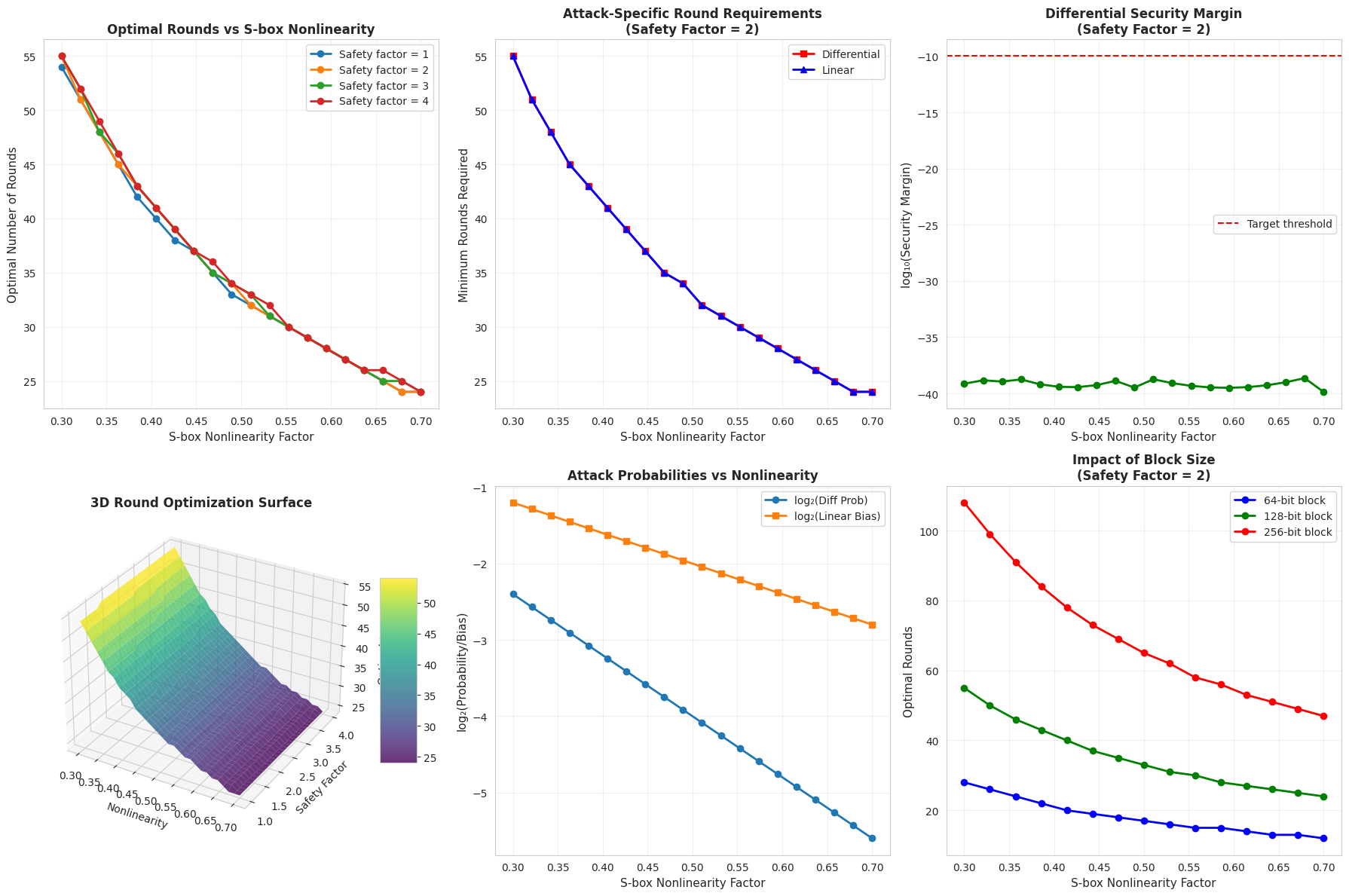
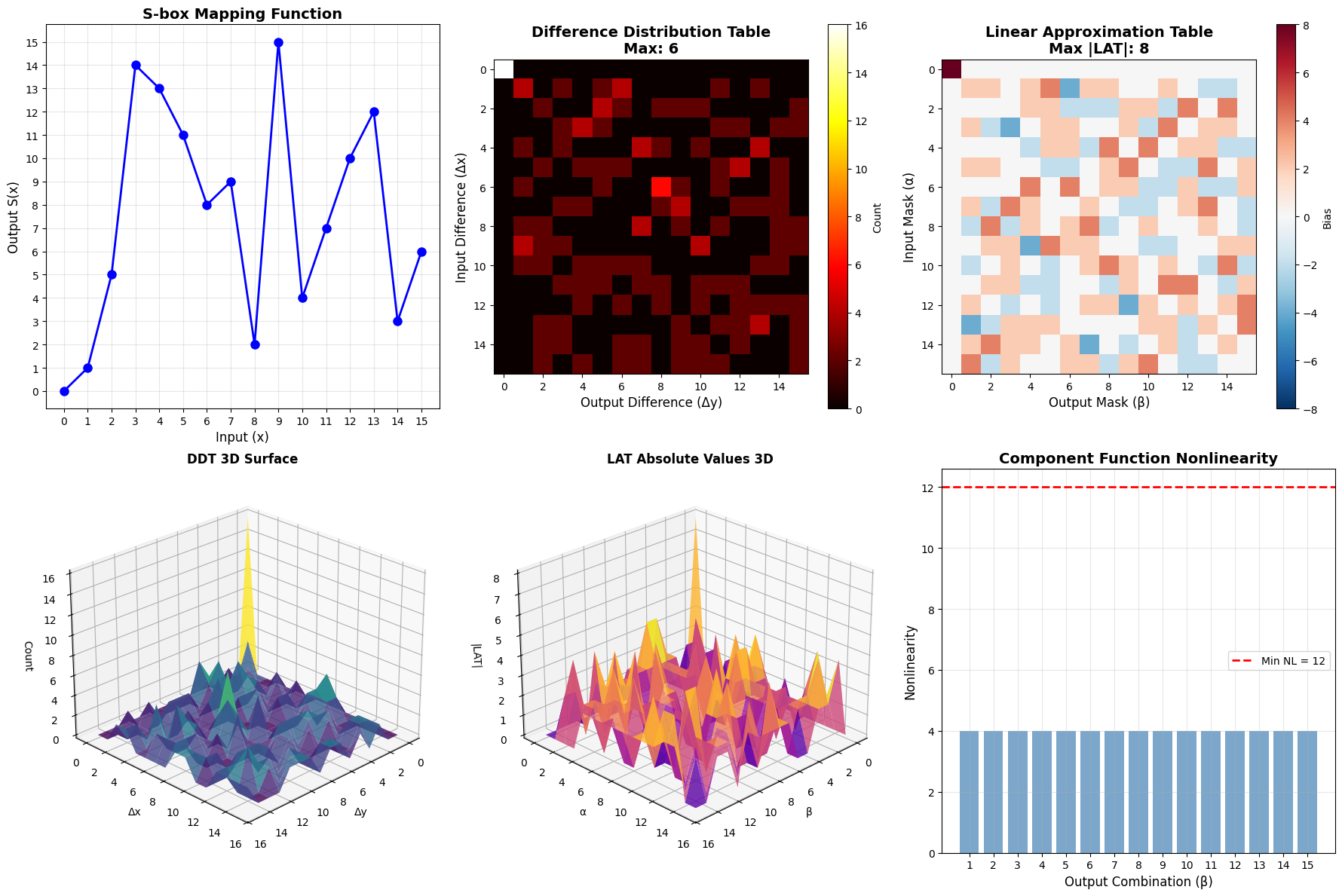
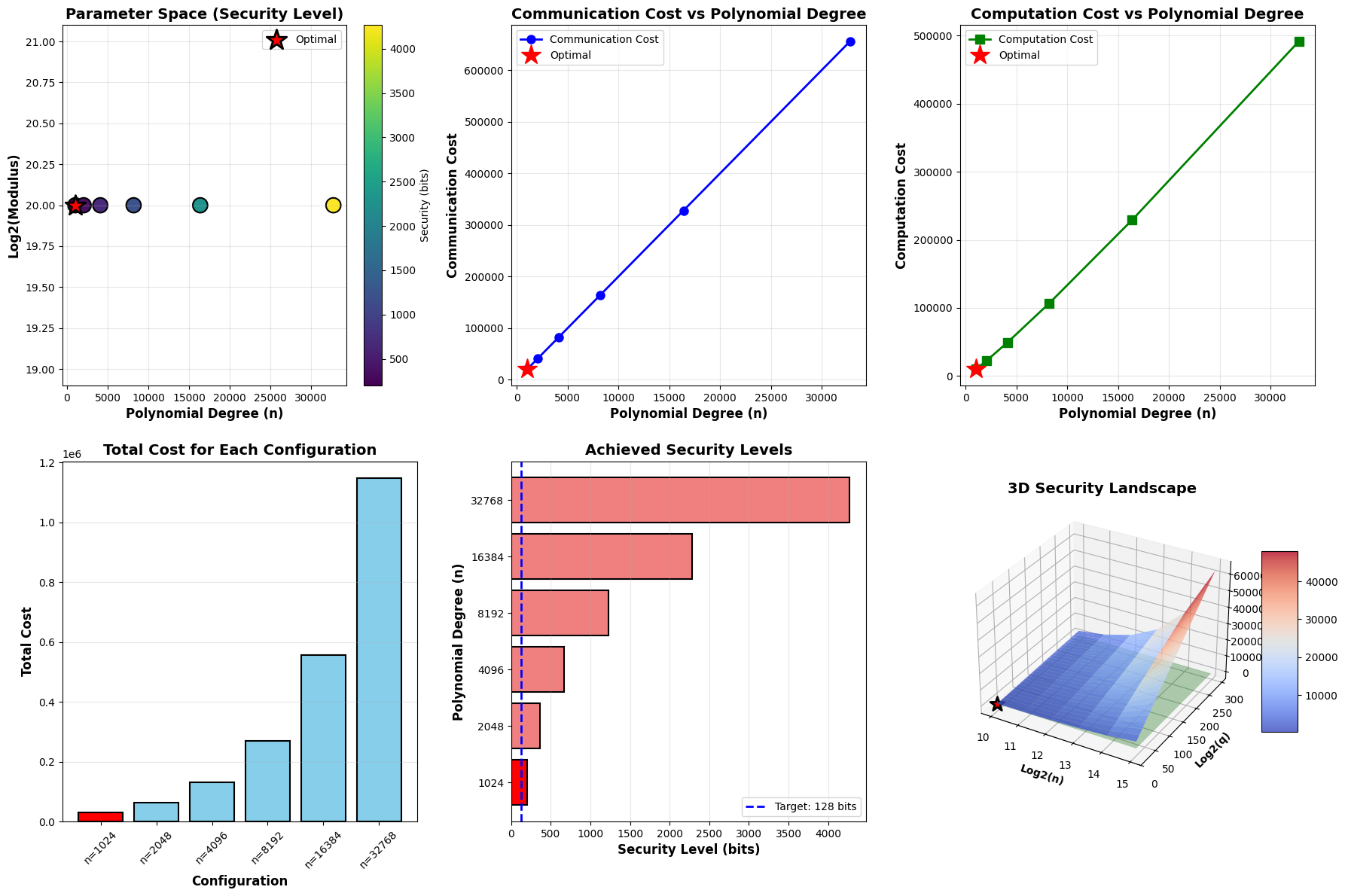
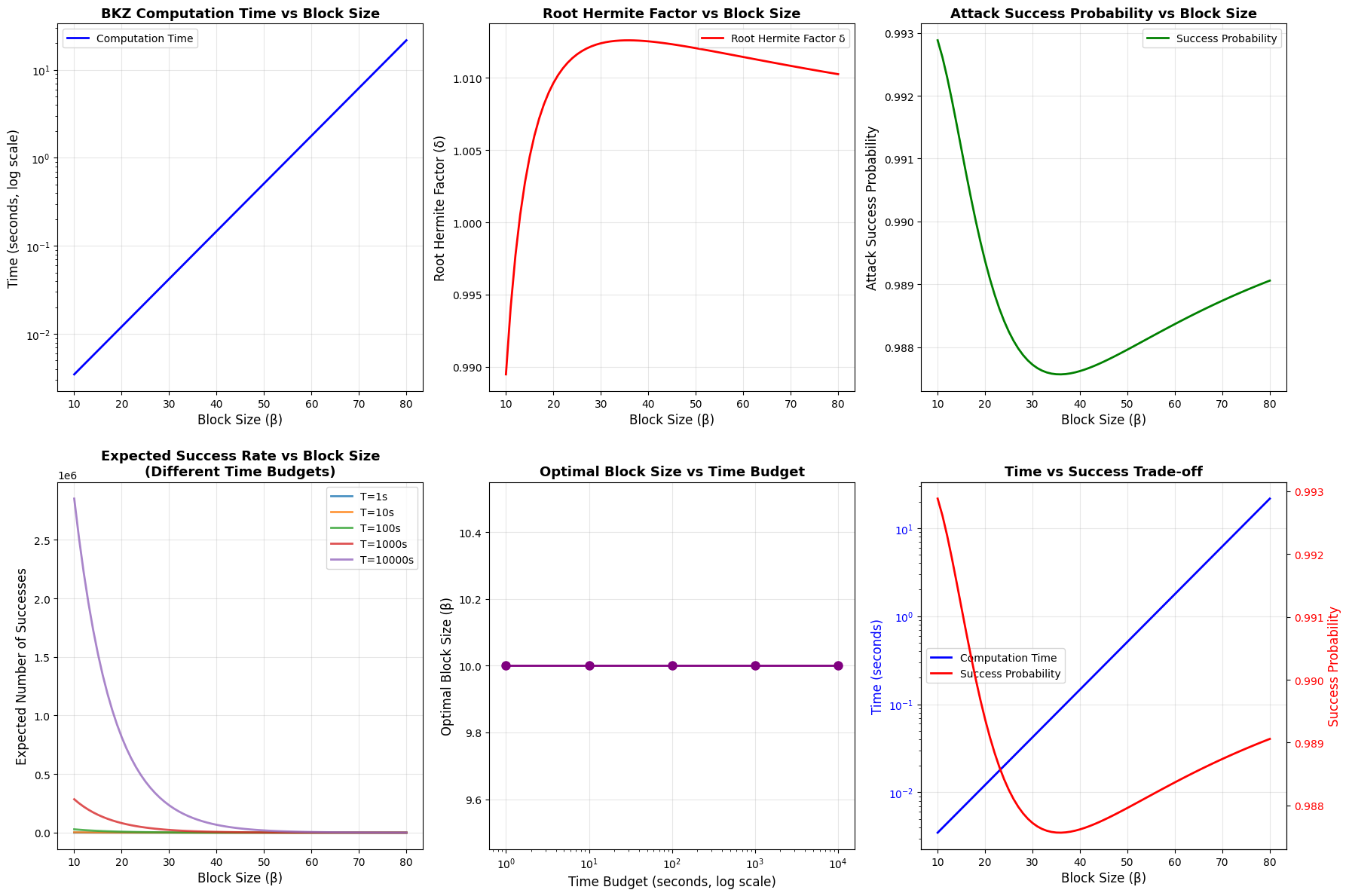
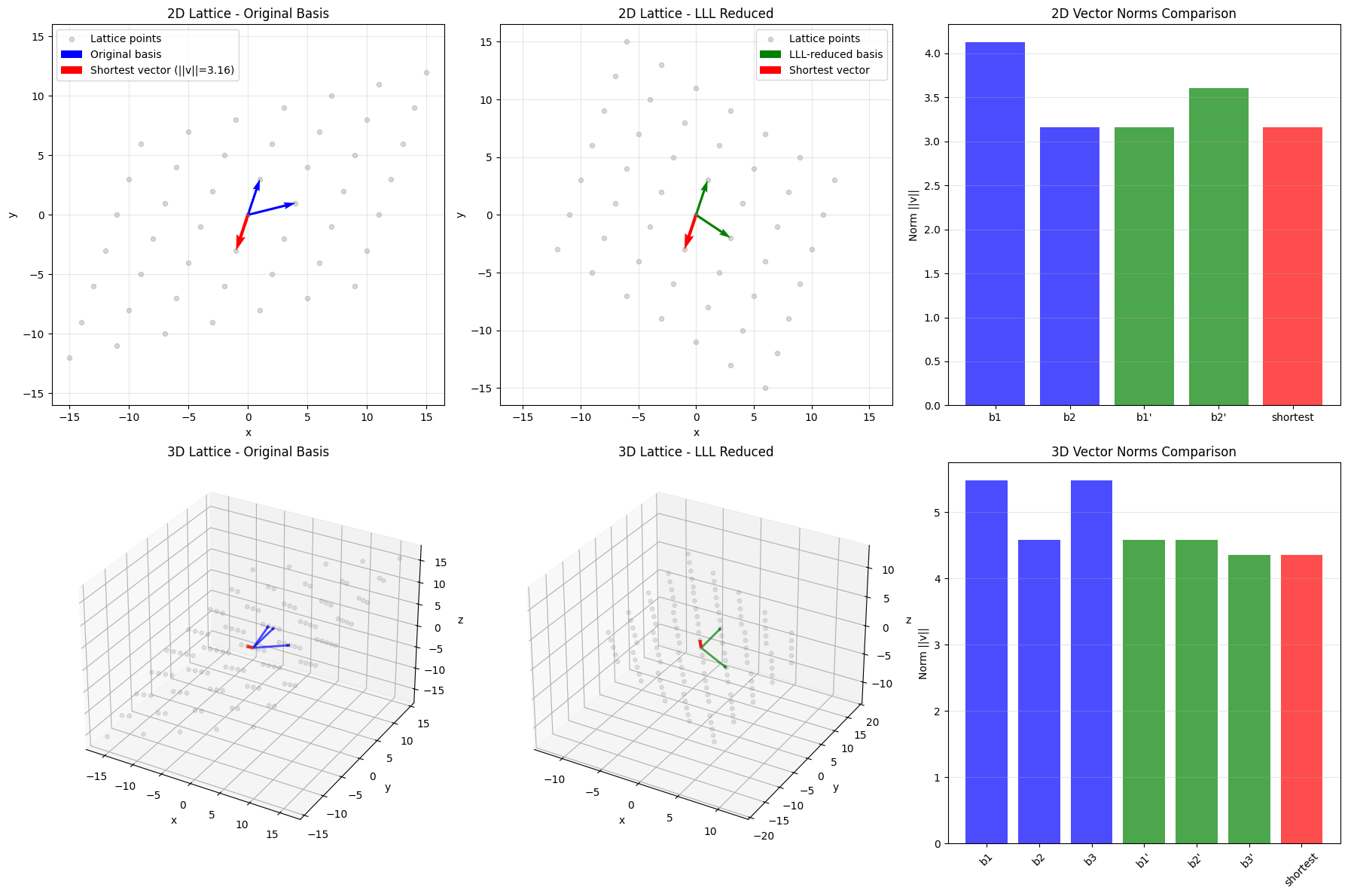
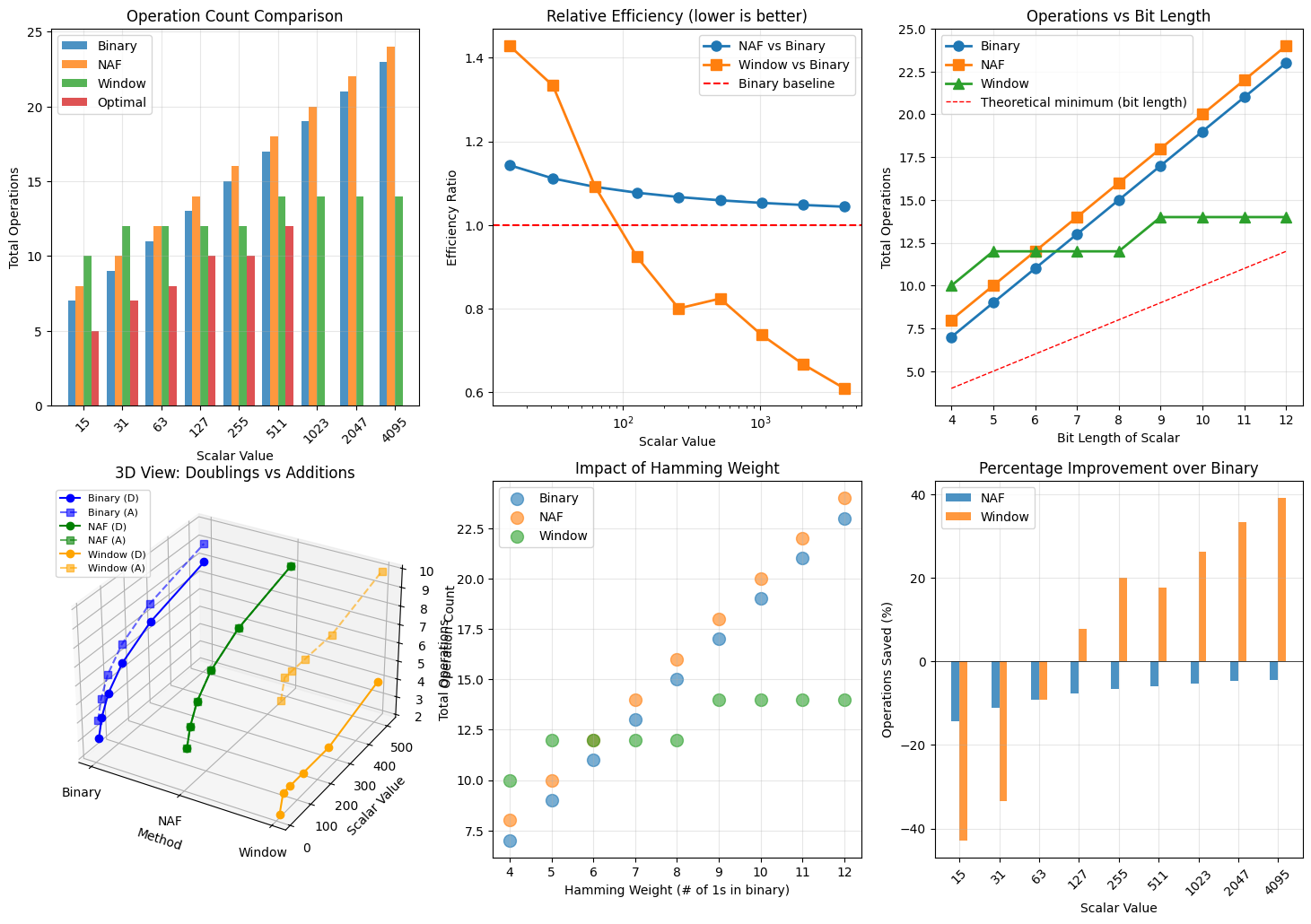
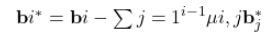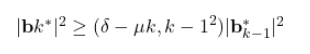1
2
3
4
5
6
7
8
9
10
11
12
13
14
15
16
17
18
19
20
21
22
23
24
25
26
27
28
29
30
31
32
33
34
35
36
37
38
39
40
41
42
43
44
45
46
47
48
49
50
51
52
53
54
55
56
57
58
59
60
61
62
63
64
65
66
67
68
69
70
71
72
73
74
75
76
77
78
79
80
81
82
83
84
85
86
87
88
89
90
91
92
93
94
95
96
97
98
99
100
101
102
103
104
105
106
107
108
109
110
111
112
113
114
115
116
117
118
119
120
121
122
123
124
125
126
127
128
129
130
131
132
133
134
135
136
137
138
139
140
141
142
143
144
145
146
147
148
149
150
151
152
153
154
155
156
157
158
159
160
161
162
163
164
165
166
167
168
169
170
171
172
173
174
175
176
177
178
179
180
181
182
183
184
185
186
187
188
189
190
191
192
193
194
195
196
197
198
199
200
201
202
203
204
205
206
207
208
209
210
211
212
213
214
215
216
217
218
219
220
221
222
223
224
225
226
227
228
229
230
231
232
233
234
235
236
237
238
239
240
241
242
243
244
245
246
247
248
249
250
251
252
253
254
255
256
257
258
259
260
261
262
263
264
265
266
267
268
269
270
271
272
273
274
275
276
277
278
279
280
281
282
283
284
285
286
287
288
289
290
291
292
293
294
295
296
297
298
299
300
301
302
303
304
305
306
307
308
309
310
311
312
313
314
315
316
317
318
319
320
321
322
323
324
325
326
327
| import numpy as np
import matplotlib.pyplot as plt
from mpl_toolkits.mplot3d import Axes3D
from scipy.linalg import qr
import time
def babai_algorithm(basis, target):
"""
Babai's nearest plane algorithm for CVP approximation
Args:
basis: numpy array of shape (n, n) representing lattice basis
target: numpy array of shape (n,) representing target point
Returns:
closest_point: approximation of closest lattice point
coefficients: integer coefficients
"""
Q, R = qr(basis.T)
coeffs_real = np.linalg.solve(basis.T, target)
coeffs_int = np.round(coeffs_real).astype(int)
closest_point = basis.T @ coeffs_int
return closest_point, coeffs_int
def enumerate_cvp(basis, target, search_radius=5):
"""
Brute force enumeration to find exact CVP solution
Args:
basis: numpy array of shape (n, n)
target: numpy array of shape (n,)
search_radius: how far to search in coefficient space
Returns:
best_point: exact closest lattice point
best_coeffs: corresponding coefficients
min_distance: minimum distance found
"""
n = basis.shape[1]
best_distance = float('inf')
best_point = None
best_coeffs = None
ranges = [range(-search_radius, search_radius + 1) for _ in range(n)]
import itertools
for coeffs in itertools.product(*ranges):
coeffs_array = np.array(coeffs)
lattice_point = basis.T @ coeffs_array
distance = np.linalg.norm(target - lattice_point)
if distance < best_distance:
best_distance = distance
best_point = lattice_point
best_coeffs = coeffs_array
return best_point, best_coeffs, best_distance
def generate_lattice_points(basis, range_limit=5):
"""
Generate lattice points for visualization
"""
n = basis.shape[1]
points = []
import itertools
ranges = [range(-range_limit, range_limit + 1) for _ in range(n)]
for coeffs in itertools.product(*ranges):
coeffs_array = np.array(coeffs)
point = basis.T @ coeffs_array
points.append(point)
return np.array(points)
print("=" * 60)
print("Closest Vector Problem (CVP) Solver")
print("=" * 60)
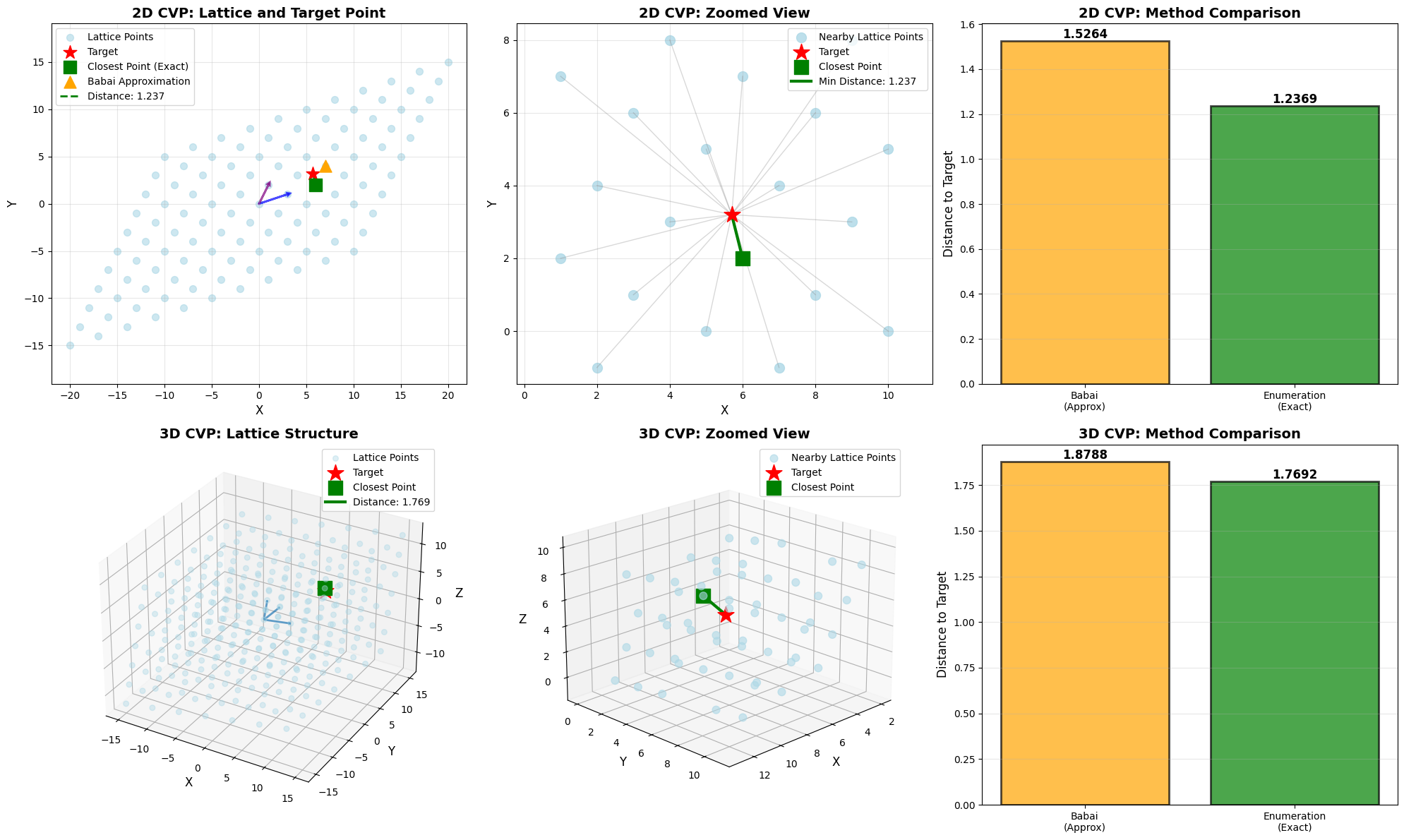
basis_2d = np.array([[3, 1],
[1, 2]])
target_2d = np.array([5.7, 3.2])
print("\nLattice Basis B:")
print(basis_2d)
print(f"\nTarget Point t: {target_2d}")
print("\n" + "=" * 60)
print("Method 1: Babai's Nearest Plane Algorithm (Fast Approximation)")
print("=" * 60)
start_time = time.time()
babai_point, babai_coeffs = babai_algorithm(basis_2d, target_2d)
babai_time = time.time() - start_time
babai_distance = np.linalg.norm(target_2d - babai_point)
print(f"Coefficients: {babai_coeffs}")
print(f"Closest Point (approx): {babai_point}")
print(f"Distance: {babai_distance:.6f}")
print(f"Computation Time: {babai_time:.6f} seconds")
print("\n" + "=" * 60)
print("Method 2: Exhaustive Enumeration (Exact Solution)")
print("=" * 60)
start_time = time.time()
exact_point, exact_coeffs, exact_distance = enumerate_cvp(basis_2d, target_2d, search_radius=10)
enum_time = time.time() - start_time
print(f"Coefficients: {exact_coeffs}")
print(f"Closest Point (exact): {exact_point}")
print(f"Distance: {exact_distance:.6f}")
print(f"Computation Time: {enum_time:.6f} seconds")
print("\n" + "=" * 60)
print("3D Lattice Example")
print("=" * 60)
basis_3d = np.array([[4, 1, 0],
[1, 3, 1],
[0, 1, 3]])
target_3d = np.array([7.5, 5.2, 4.8])
print("\n3D Lattice Basis B:")
print(basis_3d)
print(f"\nTarget Point t: {target_3d}")
print("\nBabai's Algorithm (3D):")
babai_point_3d, babai_coeffs_3d = babai_algorithm(basis_3d, target_3d)
babai_distance_3d = np.linalg.norm(target_3d - babai_point_3d)
print(f"Coefficients: {babai_coeffs_3d}")
print(f"Closest Point: {babai_point_3d}")
print(f"Distance: {babai_distance_3d:.6f}")
print("\nExhaustive Enumeration (3D):")
start_time = time.time()
exact_point_3d, exact_coeffs_3d, exact_distance_3d = enumerate_cvp(basis_3d, target_3d, search_radius=5)
enum_time_3d = time.time() - start_time
print(f"Coefficients: {exact_coeffs_3d}")
print(f"Closest Point: {exact_point_3d}")
print(f"Distance: {exact_distance_3d:.6f}")
print(f"Computation Time: {enum_time_3d:.6f} seconds")
print("\n" + "=" * 60)
print("Generating Visualizations...")
print("=" * 60)
lattice_points_2d = generate_lattice_points(basis_2d, range_limit=5)
lattice_points_3d = generate_lattice_points(basis_3d, range_limit=3)
fig = plt.figure(figsize=(20, 12))
ax1 = fig.add_subplot(2, 3, 1)
ax1.scatter(lattice_points_2d[:, 0], lattice_points_2d[:, 1],
c='lightblue', s=50, alpha=0.6, label='Lattice Points')
ax1.scatter(target_2d[0], target_2d[1],
c='red', s=200, marker='*', label='Target', zorder=5)
ax1.scatter(exact_point[0], exact_point[1],
c='green', s=150, marker='s', label='Closest Point (Exact)', zorder=5)
ax1.scatter(babai_point[0], babai_point[1],
c='orange', s=150, marker='^', label='Babai Approximation', zorder=5)
origin = np.array([0, 0])
ax1.arrow(origin[0], origin[1], basis_2d[0, 0], basis_2d[1, 0],
head_width=0.3, head_length=0.3, fc='blue', ec='blue', linewidth=2, alpha=0.7)
ax1.arrow(origin[0], origin[1], basis_2d[0, 1], basis_2d[1, 1],
head_width=0.3, head_length=0.3, fc='purple', ec='purple', linewidth=2, alpha=0.7)
ax1.plot([target_2d[0], exact_point[0]], [target_2d[1], exact_point[1]],
'g--', linewidth=2, label=f'Distance: {exact_distance:.3f}')
ax1.set_xlabel('X', fontsize=12)
ax1.set_ylabel('Y', fontsize=12)
ax1.set_title('2D CVP: Lattice and Target Point', fontsize=14, fontweight='bold')
ax1.legend(fontsize=10)
ax1.grid(True, alpha=0.3)
ax1.axis('equal')
ax2 = fig.add_subplot(2, 3, 2)
nearby_points = lattice_points_2d[
(np.abs(lattice_points_2d[:, 0] - target_2d[0]) < 5) &
(np.abs(lattice_points_2d[:, 1] - target_2d[1]) < 5)
]
ax2.scatter(nearby_points[:, 0], nearby_points[:, 1],
c='lightblue', s=100, alpha=0.8, label='Nearby Lattice Points')
ax2.scatter(target_2d[0], target_2d[1],
c='red', s=300, marker='*', label='Target', zorder=5)
ax2.scatter(exact_point[0], exact_point[1],
c='green', s=200, marker='s', label='Closest Point', zorder=5)
for point in nearby_points:
distance = np.linalg.norm(point - target_2d)
ax2.plot([target_2d[0], point[0]], [target_2d[1], point[1]],
'gray', alpha=0.3, linewidth=1)
ax2.plot([target_2d[0], exact_point[0]], [target_2d[1], exact_point[1]],
'g-', linewidth=3, label=f'Min Distance: {exact_distance:.3f}')
ax2.set_xlabel('X', fontsize=12)
ax2.set_ylabel('Y', fontsize=12)
ax2.set_title('2D CVP: Zoomed View', fontsize=14, fontweight='bold')
ax2.legend(fontsize=10)
ax2.grid(True, alpha=0.3)
ax2.axis('equal')
ax3 = fig.add_subplot(2, 3, 3)
methods = ['Babai\n(Approx)', 'Enumeration\n(Exact)']
distances = [babai_distance, exact_distance]
colors = ['orange', 'green']
bars = ax3.bar(methods, distances, color=colors, alpha=0.7, edgecolor='black', linewidth=2)
for i, (bar, dist) in enumerate(zip(bars, distances)):
height = bar.get_height()
ax3.text(bar.get_x() + bar.get_width()/2., height,
f'{dist:.4f}',
ha='center', va='bottom', fontsize=12, fontweight='bold')
ax3.set_ylabel('Distance to Target', fontsize=12)
ax3.set_title('2D CVP: Method Comparison', fontsize=14, fontweight='bold')
ax3.grid(True, alpha=0.3, axis='y')
ax4 = fig.add_subplot(2, 3, 4, projection='3d')
ax4.scatter(lattice_points_3d[:, 0], lattice_points_3d[:, 1], lattice_points_3d[:, 2],
c='lightblue', s=30, alpha=0.4, label='Lattice Points')
ax4.scatter(target_3d[0], target_3d[1], target_3d[2],
c='red', s=300, marker='*', label='Target', zorder=5)
ax4.scatter(exact_point_3d[0], exact_point_3d[1], exact_point_3d[2],
c='green', s=200, marker='s', label='Closest Point', zorder=5)
ax4.plot([target_3d[0], exact_point_3d[0]],
[target_3d[1], exact_point_3d[1]],
[target_3d[2], exact_point_3d[2]],
'g-', linewidth=3, label=f'Distance: {exact_distance_3d:.3f}')
origin_3d = np.array([0, 0, 0])
for i in range(3):
ax4.quiver(origin_3d[0], origin_3d[1], origin_3d[2],
basis_3d[0, i], basis_3d[1, i], basis_3d[2, i],
arrow_length_ratio=0.1, linewidth=2, alpha=0.7)
ax4.set_xlabel('X', fontsize=12)
ax4.set_ylabel('Y', fontsize=12)
ax4.set_zlabel('Z', fontsize=12)
ax4.set_title('3D CVP: Lattice Structure', fontsize=14, fontweight='bold')
ax4.legend(fontsize=10)
ax4.grid(True, alpha=0.3)
ax5 = fig.add_subplot(2, 3, 5, projection='3d')
nearby_points_3d = lattice_points_3d[
(np.abs(lattice_points_3d[:, 0] - target_3d[0]) < 6) &
(np.abs(lattice_points_3d[:, 1] - target_3d[1]) < 6) &
(np.abs(lattice_points_3d[:, 2] - target_3d[2]) < 6)
]
ax5.scatter(nearby_points_3d[:, 0], nearby_points_3d[:, 1], nearby_points_3d[:, 2],
c='lightblue', s=60, alpha=0.6, label='Nearby Lattice Points')
ax5.scatter(target_3d[0], target_3d[1], target_3d[2],
c='red', s=300, marker='*', label='Target', zorder=5)
ax5.scatter(exact_point_3d[0], exact_point_3d[1], exact_point_3d[2],
c='green', s=200, marker='s', label='Closest Point', zorder=5)
ax5.plot([target_3d[0], exact_point_3d[0]],
[target_3d[1], exact_point_3d[1]],
[target_3d[2], exact_point_3d[2]],
'g-', linewidth=3)
ax5.set_xlabel('X', fontsize=12)
ax5.set_ylabel('Y', fontsize=12)
ax5.set_zlabel('Z', fontsize=12)
ax5.set_title('3D CVP: Zoomed View', fontsize=14, fontweight='bold')
ax5.legend(fontsize=10)
ax5.view_init(elev=20, azim=45)
ax5.grid(True, alpha=0.3)
ax6 = fig.add_subplot(2, 3, 6)
methods_3d = ['Babai\n(Approx)', 'Enumeration\n(Exact)']
distances_3d = [babai_distance_3d, exact_distance_3d]
colors_3d = ['orange', 'green']
bars_3d = ax6.bar(methods_3d, distances_3d, color=colors_3d, alpha=0.7, edgecolor='black', linewidth=2)
for i, (bar, dist) in enumerate(zip(bars_3d, distances_3d)):
height = bar.get_height()
ax6.text(bar.get_x() + bar.get_width()/2., height,
f'{dist:.4f}',
ha='center', va='bottom', fontsize=12, fontweight='bold')
ax6.set_ylabel('Distance to Target', fontsize=12)
ax6.set_title('3D CVP: Method Comparison', fontsize=14, fontweight='bold')
ax6.grid(True, alpha=0.3, axis='y')
plt.tight_layout()
plt.savefig('cvp_analysis.png', dpi=300, bbox_inches='tight')
plt.show()
print("\nVisualization complete!")
print("=" * 60)
|